Temperature-Composition Diagram for Immiscible Liquids
Temperature-Composition Diagram for Immiscible Liquids
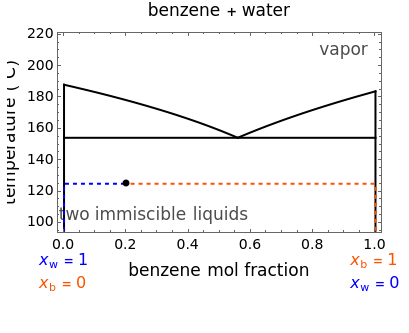
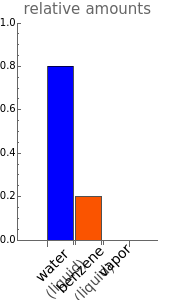
The phase diagram for two immiscible liquids, benzene and water, shows that either liquid can be in equilibrium with a vapor mixture of water and benzene. At a given pressure, only at one temperature can all three phases coexist in equilibrium (liquid water, liquid benzene, and a vapor phase containing both components). At this temperature, when heat is added, one of the liquid phases must completely evaporate before the temperature increases. A change in the total pressure causes a change in the saturation temperatures for both components.You can add heat to a mixture to observe how the amounts in each phase change; the bar graph shows the relative amounts of liquid water (blue), liquid benzene (orange), and the vapor containing both components (green).