Slurry Reactor with Declining Catalyst Activity
Slurry Reactor with Declining Catalyst Activity
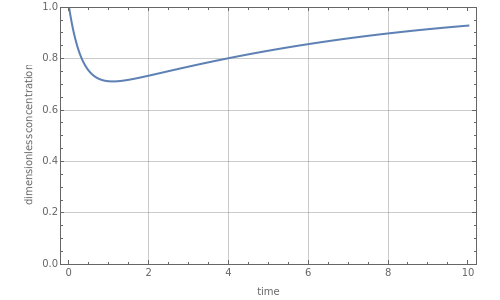
Consider a CSTR (continuous stirred-tank reactor) with a slurried catalyst where a first-order reaction is taking place. The catalyst is subject to deactivation which follows a first-order law. Thus the catalyst activity, , obeys the governing equation =-α with .
AB
k
→
α
dα
dt
k
d
α(0)=1
This Demonstration plots the dimensionless concentration, , versus time for user-set values of the reactor's residence time, catalyst deactivation, and first-order reaction rate constants. Here the feed concentration of species to the reactor and its initial concentration in the reactor are both taken equal to . You can observe the behavior of catalyst activity versus time, which depends solely on the catalyst deactivation rate constant. It is found that the dimensionless concentration, , goes through a minimum when there is deactivation; it reaches a steady state value when =0 (the case of no deactivation) as displayed in the second snapshot.
C
C
0
A
C
0
C
C
0
k
d