Single Solute Batch Two-Phase Extraction
Single Solute Batch Two-Phase Extraction
A single solute, , is being transferred from a feed into an immiscible fresh solvent, , in a batch two-phase extraction system. This Demonstration displays the dynamic approach to equilibrium for values of the linear equilibrium distribution constant, the solvent to feed-charge ratio, and the mass-transfer capacity coefficient, all to be set by the user.
A
S
The solute balances in the feed and solvent are given by
dX
dt
K
L
*
X
dY
dt
K
L
*
X
V
L
V
G
where a is the mass transfer capacity coefficient expressed in , and are the concentrations of solute in the feed and solvent (), respectively, and is the feed to solvent charge ratio. Assuming a linear equilibrium relationship gives , where is the equilibrium distribution constant.
K
L
-1
hr
X
Y
A
kg
3
m
V
L
V
G
Y=m
*
X
m
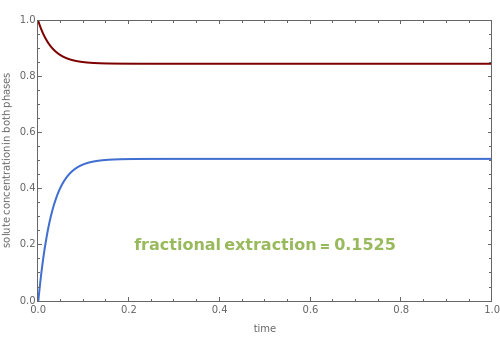
Values of the fractional extraction, defined as , are also determined. The red and blue curves correspond to the solute concentrations in the feed and solvent phases, respectively.
Z=-
X
t=0
X
t=∞
X
t=0
Observe that both a and have an effect on the system time constant. Indeed, the steady-state concentration is reached faster when, for instance, you increase a.
K
L
V
L
V
G
K
L
As expected, a has no effect on the values of the fractional extraction. On the other hand, the fractional extraction is affected by the choice of and .
K
L
m
V
L
V
G