Shell Structure of Noble Gas Atoms
Shell Structure of Noble Gas Atoms
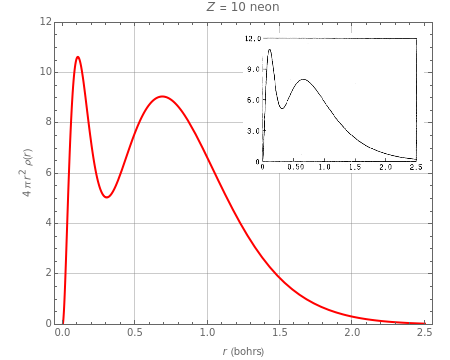
Accurate Hartree–Fock computations were carried out long ago on all the atoms in the periodic table [1]. From these results, Wang and Parr [2] have plotted radial distribution functions (RDFs) , where is the total electron density, for the noble gas atoms He (), Ne (), Ar (), Kr (), and Xe (). These are shown as insets in the graphics. The shell structure of the electron distributions is evident, and can be identified with the K, L, M, N, and O shells, corresponding to principal quantum numbers 1 to 5, respectively.
D(r)=4πρ(r)
2
r
ρ(r)
Z=2
Z=10
Z=18
Z=36
Z=54
This Demonstration proposes a simple analytic approximation for the RDFs, based on a superposition of simple Slater-type atomic orbitals (STOs). An STO has the form , where is a constant, is the nuclear charge, is an empirical shielding constant, and is an effective principal quantum number equal to 1, 2, 3, 3.7, and 4 for the principal quantum numbers , respectively. The optimal shielding constants can be determined from Slater's rules [3] but, in this application, we treat them as adjustable constants. We treat the spherically symmetric complete shells in noble gas atoms containing 2, 8, or 18 electrons as if they were single -orbitals, normalized to , the number of electrons in a shell. Thus, the contribution to the RDF from a single shell with quantum number has the form (r)=const2π(r)=Γ(2+1). The total RDF is the sum of up to five shells, with the appropriate values of , , , and (for Xe, we replace in the fifth term by .25).
ϕ{r}=c-1
*
n
r
-r(Z-σ)/
*
n
e
c
Z
σ
*
n
n=1,2,3,4,5
ns
N=2,8,18
n
D
n
2
r
2
ϕ
n
2+1
*
n
N
2Z-2σ
*
n
2
*
n
r
-2r(Z-σ)/
*
n
e
*
n
Z
N
*
n
σ
N
The object is to find values of , , , , and for each of the noble gas atoms that best reproduces the appearance of the exact RDFs. Quantitative accuracy is not attainable, particularly for Ar, Kr, and Xe, unless more adjustable parameters are introduced, say modified values for and .
σ(K)
σ(L)
σ(N)
σ(M)
σ(O)
N
*
n