Separation of a Reactive Ternary Mixture with a Distillation Column
Separation of a Reactive Ternary Mixture with a Distillation Column
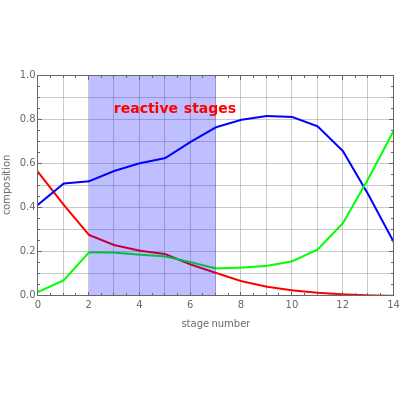
Consider a ternary mixture of components , , and with relative volatilities =5 and =3. This mixture is subject to an equilibrium-limited chemical reaction with reaction rate , where the equilibrium constant =1.5. This mixture is fed to a reactive distillation column such that the total number of plates is equal to 14, the feed stage location is stage number 5, the reactive stages are from stage 2 to stage 7, and the feed composition is equimolar in and (i.e., the feed is composed of 40% mole , 40% mole , and 20% mole ). The feed flow rate is chosen equal to 100 kmol/hr. For simplicity, the constant molal overflow (CMO) assumption is made and heat effects are neglected.
A
B
C
α
13
α
23
A+B⇌C
r=k-
x
A
x
B
x
C
K
eq
K
eq
A
B
A
B
C
This Demonstration shows two profiles: (1) the composition versus plate number for components , , and (in red, blue, and green, respectively) with the reactive zone shown in light blue and (2) a ternary diagram where the composition of versus the composition of is in % mole. In the second profile, the feed composition is shown by a magenta dot and the reactive stages (stages 2 to 7) are displayed in blue.
A
B
C
A
B
You can set the values of the Damköhler number, , the reflux ratio, and the reboil ratio. When , one recovers the case where no reaction is taking place. If is very large, the simulation represents a situation close to reaction equilibrium (i.e., ).
Da
Da=0
Da
r=0
One snapshot shows a case where the reflux ratio is very high, a situation close to the total reflux operation. For such a situation, the bottom is almost pure and the distillate contain very little . The distillation flow is very small and the distillate stream can be recycled if desired. Thus, for this specific operation, one can produce , if is the desired reaction product, and convert both reactants and with a single piece of equipment.
C
C
C
C
A
B