Relativistic Energy Levels for Hydrogen Atom
Relativistic Energy Levels for Hydrogen Atom
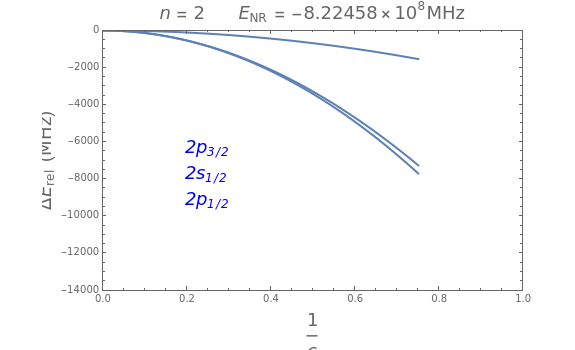
In nonrelativistic quantum mechanics, the energy levels of the hydrogen atom are given by the formula of Bohr and Schrödinger, =-, expressed in hartrees (assuming the appropriate correction for the reduced mass of the electron). The energy depends only on the principal quantum number and is -fold degenerate (including electron spin). In Dirac's relativistic theory, this degeneracy is partially resolved and the energy is found to depend as well on the angular-momentum quantum number . To second order in the fine-structure constant , the hydrogen energy levels are given by =-1+-. In Dirac's theory, levels such as and remain degenerate. The discovery of the Lamb shift showed that these two levels were actually split by 1057.8 MHz. This was a major stimulus for the development of quantum electrodynamics in the 1950s. The Lamb shift, significant only for (s-states), raises the energy by approximately . The relativistic and radiative correction to hydrogen energy levels can therefore be written , to third order in . In this Demonstration, you can conceptually vary the fine-structure constant from 0 to its actual value, or equivalently the speed of light from to 1 (meaning m/s), to show the transition from nonrelativistic to relativistic energies for quantum numbers , and . The energies are expressed in MHz (1 hartree = MHz).
E
n
1
2
2
n
n=1,2,3,…
2
2
n
j
α=/ℏc≈1/137
2
e
E
nj
1
2
2
n
2
α
2
n
n
j+1/2
3
4
2
s
1/2
2
p
1/2
l = 0
13
4
3
α
3
n
Δ=--+
E
rel
2
α
2
4
n
n
j+1/2
3
4
13
4
3
α
3
n
δ
l,0
α
c
∞
3×
8
10
n=1,2
3
6.57966×
9
10
A checkbox lets you view a schematic diagram of hydrogen energy levels for various levels of the theory. This includes also the hyperfine structure, from interaction between electron and nuclear magnetic moments. The transition in the level gives rise to the famous 1420 MHz (21 cm) microwave radiation in radio astronomy.
F=0←F=1
1
s
1/2