Pauling Electronegativity and Dipole Moment
Pauling Electronegativity and Dipole Moment
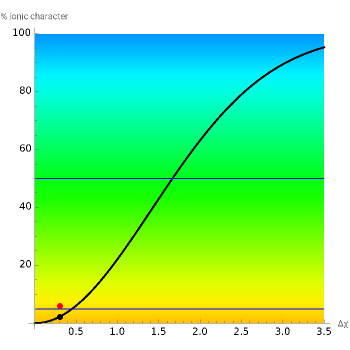
This Demonstration shows the relationship between the dipole moment of molecules and Pauling's electronegativity. Pauling defined electronegativity [1] and proposed the following relation: . This is an empirical approximation and sometimes is not accurate. More recent approaches are more complex and depend on other electronic properties of the molecule [2].
%ioniccharacter=1-
1
4
2
(-)
χ
a
χ
b
e
The "experimental" window uses some experimental data and compares them with the theoretical prediction. An ionic bond does not mean necessarily 100% ionic character; anything above 50% is sufficient to classify the bond as ionic. When the bond is ionic, according to the experimental value, one element is colored red (positive charge) and the other one blue (negative charge). In the case of a polar covalent bond, a segment links both elements to indicate the electron pair shared between the two atoms. The color intensity is lighter than the ionic bond as the electronic distribution is asymmetrical [3]. Moving the "dipole" slider gives a new example with the predicted value shown.
The "theoretical" window shows the plot associated with the bonding molecular orbital distribution of two generic orbitals. Three areas are highlighted according only to the electronegativity difference: nonpolar, polar covalent and ionic bond [4].
s