Oxidation of Naphthalene in a Tubular Flow Reactor
Oxidation of Naphthalene in a Tubular Flow Reactor
Consider the oxidation of naphthalene in a tubular flow reactor with constant wall temperature, which is taken equal to the reactor's inlet temperature.
The governing equations (mass and energy balance equations) are
u=r
dp
dz
-MΠ
ρ
b
ρ
g
u=r-R(T-)
dT
dz
-
ΔH
r
ρ
b
C
p
2U
C
p
T
m
r=0.208exp19.837-p
13636
T
where is the reaction rate, is the axial distance along the reactor, is a heat transfer coefficient, is the reactor's temperature, is the partial pressure of naphthalene, is the atmospheric pressure, Δ is the heat of reaction, is the molecular weight, and are the bulk and gas densities, respectively, is the heat capacity, is the tube radius, is the wall and inlet temperature, and is the linear velocity.
r
z
U
T
p
Π
H
r
M
ρ
b
ρ
g
C
p
R
T
m
u
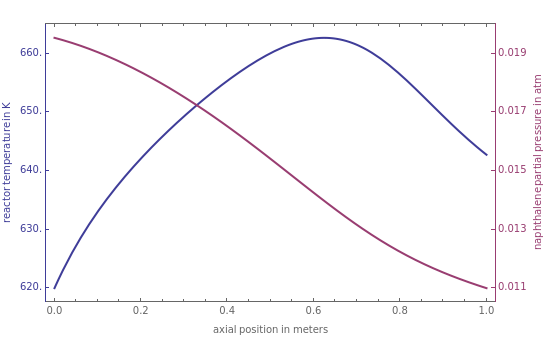
The Demonstration finds the profile of temperature and naphthalene partial pressure along the reactor for the inlet and wall temperature, which is set by the user. A small increase in the value of this parameter (e.g., from 620 to 625 K) will lead to a very dangerous situation, named thermal runaway, where the temperature, , grows exponentially, as you can see in the last snapshot.
T