Optimizing Temperatures and Volumes for Maximum Conversion to an Intermediate Reactant in a Chain of Two Continuous Stirred-Tank Reactors
Optimizing Temperatures and Volumes for Maximum Conversion to an Intermediate Reactant in a Chain of Two Continuous Stirred-Tank Reactors
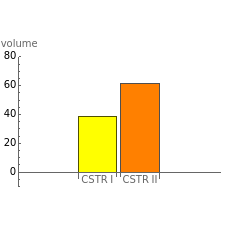
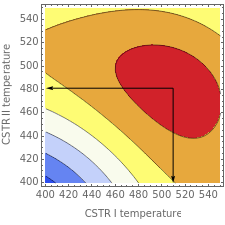
This Demonstration examines a two-stage continuous stirred-tank reactor (CSTR). Conversion to an intermediate product of two consecutive chemical reactions can be maximized by operating the system at the optimal volume and temperature for each reactor.
Consider the consecutive first-order chemical reaction where , , and represent reactants, products, and waste, = and =, where , are the rate constants, , are the Arrhenius constants, , are the activation energies, and is the temperature. The equations describing the steady state condition of the CSTR are
ABC
k
1
⟶
k
2
⟶
A
B
C
k
1
A
1
-/RT
E
1
e
k
2
A
2
-/RT
E
2
e
k
1
k
2
A
1
A
2
E
1
E
2
T
0=-q/-
c
A
n-1
c
A
n
V
n
k
1
c
A
n
0=-q/+-
c
B
n-1
c
B
n
V
n
k
1
c
A
n
k
2
c
B
n
c
A
n
c
B
n
V
n
n=1,2
q
q=2
V
1
V
2
A
0
B
0
C
0
k
1
-4000/T
e
400≤T≤550
The maximum concentration of reactant exiting the second reactor and the optimum temperature and volume of each reactor are shown for different ratios of the Arrhenius constants and activation energies.
B