Heat Capacity of Solids in the Debye Approximation
Heat Capacity of Solids in the Debye Approximation
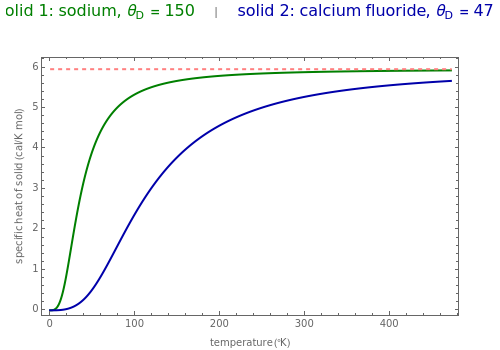
The value of the classical molar heat capacity =3R, depends on temperature. In the Debye approximation, it is given by (T)=9Tdx, where is the Debye temperature of the solid, is the absolute temperature, and is the gas constant. This Demonstration shows the variation of the specific heat of solids with temperature of representative solids according to the Debye theory.
C
V
C
V
3
R
T
θ
D
θ
D
∫
0
x
e
4
x
2
(-1)
x
e
θ
D
T
R