Exact and Variational Energies for the Hydrogen Molecular Ion and the Helium Hydride Ion
Exact and Variational Energies for the Hydrogen Molecular Ion and the Helium Hydride Ion
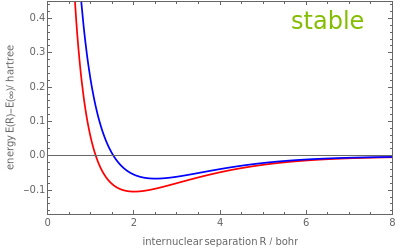
For the lowest two states of the homonuclear or heteronuclear one-electron two-center system with nuclear charges and , exact (red curves) and approximate (blue curves) energies are shown. Depending on the choice made, total energies including the nuclear repulsion ("potential energy curves"), electronic energies, or differences between the exact and approximate energies are plotted as a function of the internuclear separation. At each nuclear center, one hydrogenic 1 orbital is employed for the LCAO (Linear Combination of Atomic Orbitals) approximation. By adjusting the parameters in the exponents and of the 1 orbitals, the quality of the variational approximation can be controlled, and by changing the charge parameter one can study the stability properties of the system.
Z
a
Z
b
s
α
i
exp(-)
α
a
r
a
exp(-)
α
b
r
b
s
Z
a