Entropy Changes in Mixing Ideal Gases
Entropy Changes in Mixing Ideal Gases
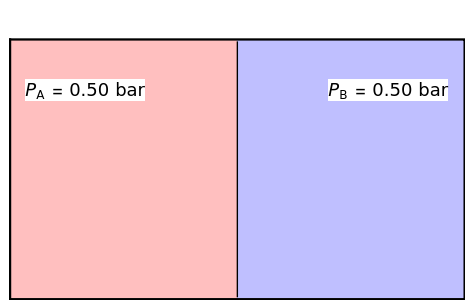
In this Demonstration two ideal gases, A and B, are mixed isothermally. After the barrier is removed, the entropy change of each gas is the same as that of a gas expanding into a vacuum. When the partial pressure decreases, entropy increases, and vice versa. If the partial pressure of a gas does not change, its entropy does not change, even when another gas is added to it. The total entropy change is the sum of the individual entropy changes. Gas A is colored red and gas B is colored blue, and when the gases mix, different shades of purple result, depending on the relative ratio of moles of each species. As the number of moles increases, the color becomes more intense.
When the initial pressures of A and B are equal, Gibbs's famous formula for the entropy of mixing applies: , where and are the mole fractions of A and B in the final mixture.
Δ=-Rln-Rln
S
mix
n
A
x
A
n
B
x
B
x
A
x
B