Electrode-Solution Interface: Distribution of Ions around an Electrode
Electrode-Solution Interface: Distribution of Ions around an Electrode
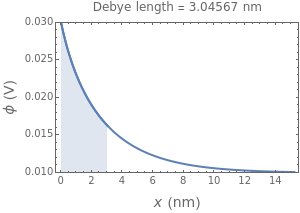
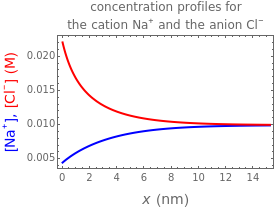
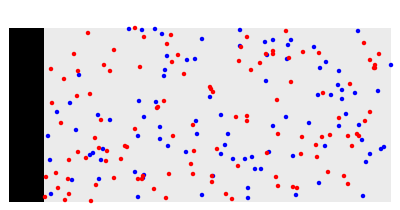
What happens if a bar of a noble metal is immersed in an electrolyte solution such as ? Due to the difference between the electric potential of the metal and the electrode-solution interface, the ions will move to equalize the total charge. This Demonstration shows the distribution of ions (cation and anion ) near an electrode surface and also the variation of potential with distance from the electrode, as functions of concentration.
NaCl
+
Na
-
Cl