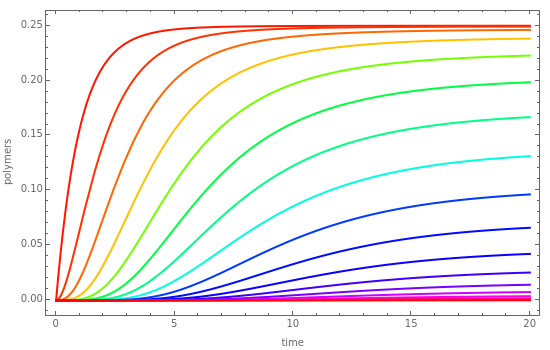
Dynamic Behavior of an Addition Polymerization in a Batch Reactor
Dynamic Behavior of an Addition Polymerization in a Batch Reactor
An activated monomer is obtained by the reaction .
M
k
0
→
P
1
Higher molecular weight polymers are obtained by the reaction .
M+
P
i-1
k
→
P
i
This Demonstration solves the system of second-order rate equations for user-set values of the activation rate constant, , and the polymerization rate constant, .
k
0
k
In addition, the monomer and polymers concentrations, up to , are plotted versus time.
P
20
The mass balance equations are
dM
dt
k
0
199
∑
i=1
P
i
d
P
n
dt
P
n-1
P
n
n=2,…,200
d
P
1
dt
k
0
P
1
You can easily see by looking at the equations that is a decreasing function of time. Thus, after a certain time and all species reach a steady-state value because the polymerization reactions will stop when the monomer is exhausted.
M
M=0