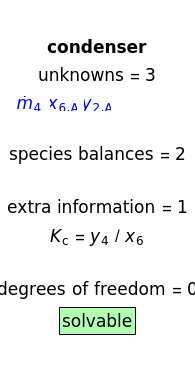Degree-of-Freedom Analysis on a Distillation Process
Degree-of-Freedom Analysis on a Distillation Process
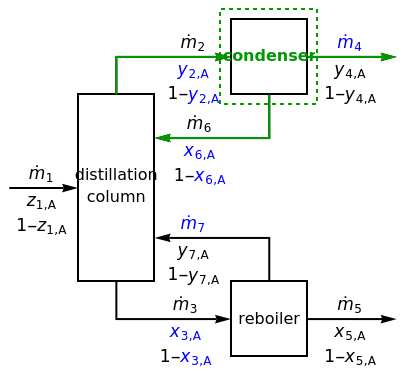
In this Demonstration, a degree-of-freedom analysis is performed on a distillation process to determine whether the system has sufficient information to solve for the unknown variables. Use buttons to select the unit on which to do mass balances (distillation column, condenser, or reboiler), or to do a balance on the overall system. When the condenser is selected, use buttons to make it a partial or a total condenser.
Set the number of unknown variables with the slider; unknown variables are colored blue on the diagram, while known variables turn black. The variables that are specified are selected randomly, and they change when you click the "new problem" button. Use buttons to select how species is represented (=1- or as ); the degree-of-freedom analysis is different for each representation.
B
z
B
z
A
z
B
An explanation of the analysis is shown at the right of the diagram. If there are zero degrees of freedom, the selected balance is solvable. For a reboiler the equilibrium ratio is a known variable, and for a partial condenser the equilibrium ratio is a known variable. Assume that temperatures and pressures are known. Mass balances and phase-equilibrium relations provide the equations used to solve the system.
K
r
K
c