Conformational Analysis of Ethane, Butane and 2-Butanol
Conformational Analysis of Ethane, Butane and 2-Butanol
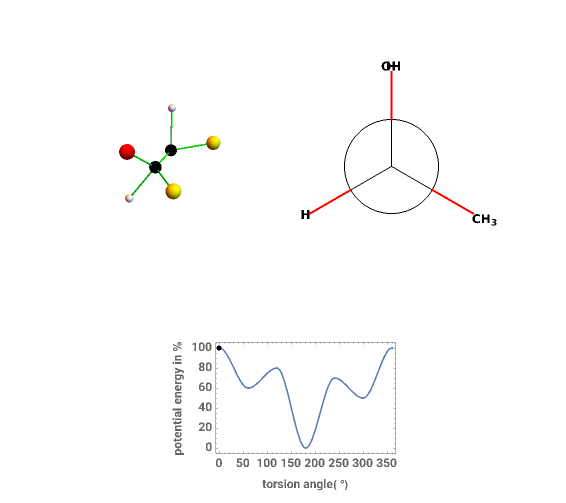
This Demonstration shows the dependence on the C-C bond torsion angle of the potential energy of three open-chain organic compounds, which results from steric effects among the various atoms or groups within the molecule.
Three representative molecules exhibiting these effects are considered: ethane, n-butane and 2-butanol. You can rotate one part of the molecule relative to the other and follow the rotation both in the 3D representation and the Newman projection along the C-C axis.
You can see the effect of the rotation on the potential energy by following the moving point on the plot; the abscissa shows the rotation angle in degrees, while the ordinate shows the potential energy as a percentage [1].