Complex Reaction with Segregation in a Semi-Continuous Reactor
Complex Reaction with Segregation in a Semi-Continuous Reactor
Consider the complex reaction scheme in a semi-continuous reactor: (desired reaction) and (undesired reaction). The governing equations in their dimensionless form are:
A+BR
k
1
→
R+BS
k
2
→
d
y
A
dθ
1
A
y
A
D
a
y
A
y
B
d
y
B
dθ
1
A
y
B
D
a
y
A
y
B
D
a
y
B
y
R
d
y
R
dθ
1
A
y
R
D
a
y
A
y
B
D
a
y
B
y
R
d
y
S
dθ
1
A
y
S
D
a
y
B
y
R
dI
dθ
1
A
I
θ
m
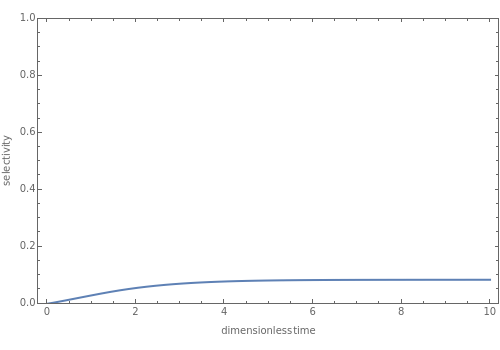
where for and for , =τ is the Damköhler number, is the reactor residence time, = is the dimensionless mixing time, is the mixing time, is dimensionless time, is the dimensionless concentration of species , is the initial concentration ratio, is the degree of segregation, and are the rate constants of the two reactions, and is the ratio of the two reaction rate constants. The initial conditions are: (0)=1,(0)=η,(0)=(0)=0, and . We define the selectivity by =+2 so that when no desired product is obtained. This Demonstration displays the selectivity as a function of the dimensionless time for various values of , , , and .
A=θ
θ<1
A=1
θ>1
D
a
k
2
-
c
A0
τ=
V
F
θ
m
τ
m
τ
τ
m
θ=
t
τ
m
y
i
i
η=
-
c
B0
-
c
A0
I
k
1
k
2
β=
k
1
k
2
y
A
y
B
y
R
y
S
I(0)=1
S
R
2
y
S
y
R
y
S
S=1
R
S
R
θ
β
η
D
a
θ
m
Lower values of (i.e., that give more of the desired product ) are obtained when:
S
R
R
1. , , and are fixed and is increased
η
D
a
θ
m
β
2. , , and are fixed and is decreased
β
η
θ
m
D
a
3. , , are fixed and and is decreased
β
D
a
θ
m
η
4. , , and are fixed and is decreased
β
η
D
a
θ
m