Bonding and Antibonding Molecular Orbitals
Bonding and Antibonding Molecular Orbitals
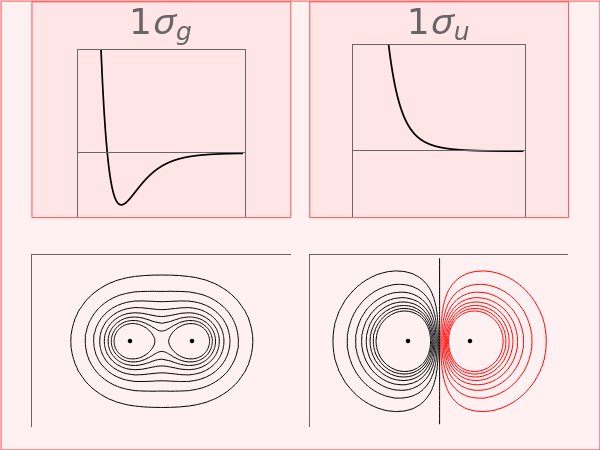
The lowest-energy bonding and antibonding molecular orbitals (MO) for a homonuclear diatomic molecule are shown, as the internuclear distance is varied. These MOs are designated 1 (or 1s) and 1 (or 1s), respectively. The red contours designate negative values of the wavefunction. In the separated atom limit (large ), the MOs approach a pair of noninteracting 1s atomic orbitals. In the united atom limit (), the 1 and 1 collapse to 1s and 2atomic orbitals, respectively. At the equilibrium internuclear distance, when the red dot is at the minimum of the energy curve, the bonding orbital is optimally effective.
R
σ
g
σ
σ
u
*
σ
R
R=0
σ
g
σ
u
p
z