Arrhenius Equations for Reaction Rate and Viscosity
Arrhenius Equations for Reaction Rate and Viscosity
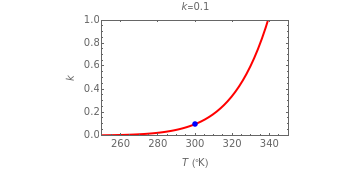
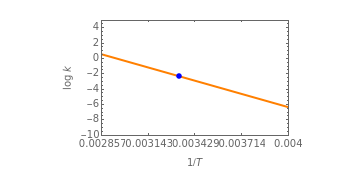
This Demonstration considers the effect of temperature on the rate of processes obeying the Arrhenius equation, specifically chemical reaction rates and fluid viscosity. We plot the reaction rate or viscosity as a function of the energy of activation. It is shown that increasing the temperature can accelerate chemical reaction rates and lower the viscosity of fluids.