Absorption and Emission of Radiation by Atoms
Absorption and Emission of Radiation by Atoms
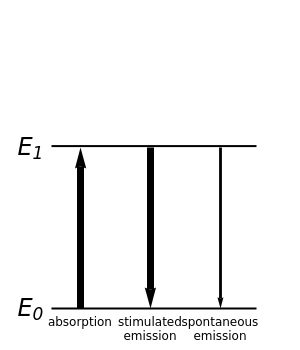
The quantum theory of radiation pictures atoms with quantized energy levels interacting with an assembly of photons of varying frequencies . This can be represented by the cubic box on the left side of the graphic. When the energy difference between two atomic states fulfills a resonance condition , three possible transition processes can occur: absorption, stimulated emission, and spontaneous emission. In the electric-dipole approximation, the transition rates for absorption and stimulated emission are given by ==ρ(ω)<0μ1>, where is the electric dipole operator and is the spectral density of the radiation field at frequency . The corresponding rate of spontaneous emission is =<0μ1>. All three radiative processes depend on the matrix element and therefore obey the same selection rules. A convenient measure of the intensity of a transition is the oscillator strength =<0μ1>, which generally lies in the range from 0 to 1. The dependence on makes spontaneous emission significant only for higher-energy transitions—in practice, only for optical frequencies and higher. This Demonstration considers the transition-energy range of 0.1 to 10 eV, corresponding to radiation wavelengths of 120 to 12000 nm, which includes the visible region 380–750 nm, as well as parts of the ultraviolet and infrared.
ω
ℏω=-
E
1
E
0
W
abs
W
stimem
4
2
π
3
2
ℏ
2
|
μ
ρ(ω)
ω
W
spontem
4
3
ω
3ℏ
3
c
2
|
<0μ1>
f
0,1
2ω
m
e
3ℏe
2
|
3
ω
The electromagnetic field is governed by the same equations as the quantum harmonic oscillator. The probabilities for annihilation and creation of a photon of frequency are given by and , respectively. The photon numbers are proportional to the spectral density , while the additional term +1 in the creation formula accounts for the phenomenon of spontaneous emission.
ω
<-1>=
n
ω
a
ω
n
ω
2
|
n
ω
<+1>=+1
n
ω
†
a
ω
n
ω
2
|
n
ω
n
ω
ρ(ω)