Raoult's Law for Ideal Solutions
Raoult's Law for Ideal Solutions
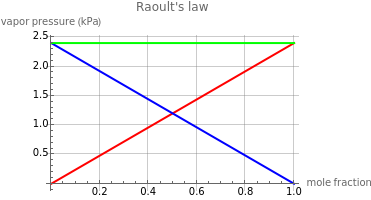
Raoult's law gives the vapor pressure of an ideal solution of two components at a constant temperature.
Use the dropdown menus to select the components in your solution. Use the sliders to specify the number of moles of each component. Component 1 is represented by the red cylinder and component 2 by the blue cylinder.
The plot illustrates Raoult's law, with vapor pressure in kPa on the axis and the mole fraction on the axis. The red and blue lines represent the partial pressures of the components, while the green line is the total vapor pressure.
y
x
Permanent Citation
Permanent Citation
Camila Amarista, Megan Tafoya
"Raoult's Law for Ideal Solutions"
http://demonstrations.wolfram.com/RaoultsLawForIdealSolutions/
Wolfram Demonstrations Project
Published: April 19, 2023