Joule-Thomson Inversion Curves for Soave-Redlich-Kwong (SRK) and Peng-Robinson (PR) Equations of State
Joule-Thomson Inversion Curves for Soave-Redlich-Kwong (SRK) and Peng-Robinson (PR) Equations of State
Throttling a real gas can sometimes result in a temperature decrease. If such is the case, the Joule–Thomson coefficient is positive. This coefficient can be written as:
μ=
∂T
∂P
H
μ=P
R
2
T
c
P
∂Z
∂T
P
where is the universal gas constant, is the constant-pressure heat capacity and is the compressibility factor. In order to estimate the value of , two equations of state are applied: the Soave–Redlich–Kwong (SRK) EoS and the Peng–Robinson (PR) EoS.
R
c
P
Z
μ
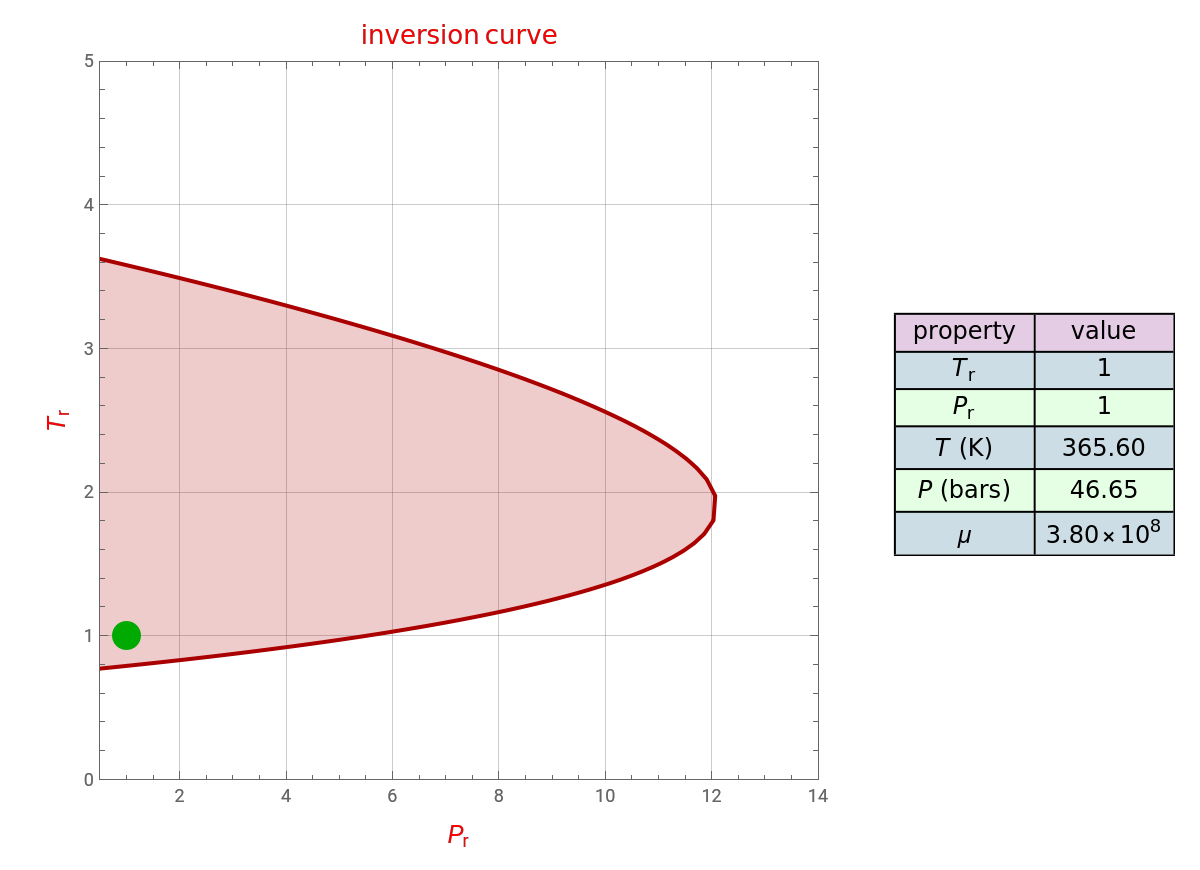
This Demonstration uses arc-length continuation to compute the Joule–Thomson inversion curve (i.e. the loci of the points, in the - plane, where ). Here we choose propylene, but the program can be modified for any other pure component. You can set the value of the reduced pressure and temperature (i.e. and , respectively). This Demonstration will mark your choice by a green dot in the - plane. If your choice corresponds to a point inside the colored area, then the value of the Joule–Thomson coefficient is positive (see Snapshot 1); otherwise (see Snapshot 2). The inversion curve and colored region are both indicated either in red (for the SRK EoS) or in blue (for the PR EoS).
P
r
T
r
μ=0
P
r
T
r
P
r
T
r
μ<0
References
References
[1] J. M. Smith, H. C. Van Ness and M. M. Abbott, Introduction to Chemical Engineering Thermodynamics, 7th ed., Boston: McGraw-Hill, 2005.
Permanent Citation
Permanent Citation
Housam Binous, Ahmed Bellagi
"Joule-Thomson Inversion Curves for Soave-Redlich-Kwong (SRK) and Peng-Robinson (PR) Equations of State"
http://demonstrations.wolfram.com/JouleThomsonInversionCurvesForSoaveRedlichKwongSRKAndPengRob/
Wolfram Demonstrations Project
Published: December 16, 2016