Reaction in an Isothermal Continuous Stirred-Tank Reactor (CSTR)
Reaction in an Isothermal Continuous Stirred-Tank Reactor (CSTR)
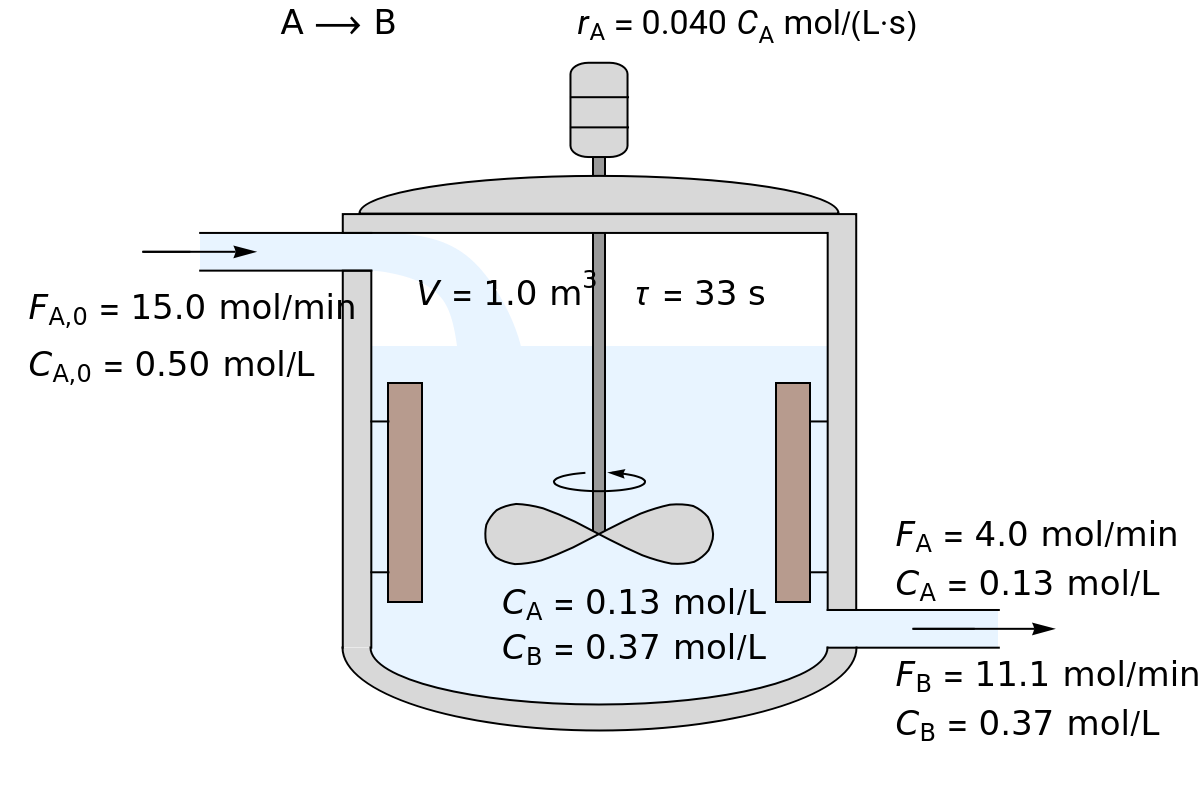
In this Demonstration, the liquid-phase reaction takes place in an isothermal, continuous stirred-tank reactor (CSTR). Use the sliders to set the feed concentration of , , the volumetric flow rate and the rate constant . Select the reaction order with respect to using the "" or "" button. The rate constant has the same numerical value when the reaction order changes, but its units are different. The figure shows the feed molar flow rate , the feed concentration , the outlet molar flow rates , and the outlet concentrations , . Note that the outlet concentrations are identical to the concentrations in the reactor. The reactor residence time is also calculated.
A⟶B
A
C
A,0
k
A
st
1
nd
2
F
A,0
C
A,0
F
A
F
B
C
A
C
B
τ=V/v
Details
Details
The constant-density, liquid-phase reaction takes place in an isothermal CSTR: , with reaction rate ,
AB
-==k
r
A
r
B
m
C
A
where is the concentration of component ,
C
A
A
r
i
i
m
A
and is the rate constant.
k
Mass balances on each component:
component : -+V=0,
A
F
A,0
F
A
r
A
component : -+V=0.
B
F
B,0
F
B
r
B
Mass balances in terms of volumetric flow rates and concentrations:
component : v-v+V=0,
A
C
A,0
C
A
r
A
component : v-v+V=0.
B
C
B,0
C
B
r
B
F
i,0
i
F
i
i
C
i,0
C
i
i
and is the volumetric flow rate, which is equal at the inlet and outlet for constant-density reactions.
v
The solution to these mass balances for first- and second-order reactions are:
first-order:
C
A
C
A,0
C
A,0
-kτ
e
C
B
C
B,0
C
A,0
C
A,0
-kτ
e
second-order:
C
A
C
A,0
-1
1
C
A,0
where is the residence time in the reactor.
τ=V/v
External Links
External Links
Permanent Citation
Permanent Citation
Neil Hendren, John L. Falconer
"Reaction in an Isothermal Continuous Stirred-Tank Reactor (CSTR)"
http://demonstrations.wolfram.com/ReactionInAnIsothermalContinuousStirredTankReactorCSTR/
Wolfram Demonstrations Project
Published: February 27, 2019