Adiabatic Flash Drum with Binary Liquid Feed
Adiabatic Flash Drum with Binary Liquid Feed
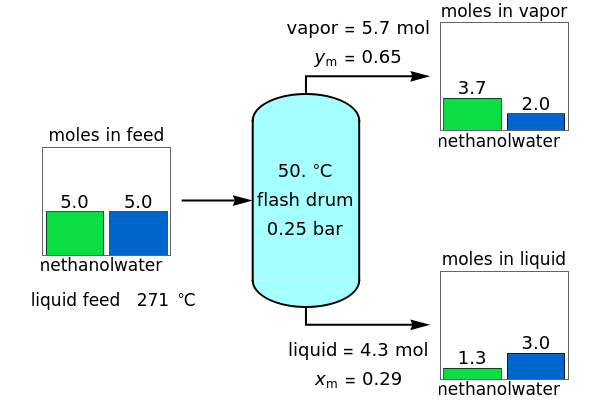
A high-pressure, hot, liquid mixture of methanol and water is fed into an adiabatic flash drum (or vapor-liquid separator). Because the flash drum pressure is below the bubble pressure, some of the liquid evaporates and the temperature decreases because energy is needed to evaporate the liquid. Thus, a vapor-liquid mixture in equilibrium exits the drum. You can vary the feed mole fraction of methanol, the feed temperature and flash drum pressure with sliders. This is a continuous process, but calculations are presented for 10 moles of feed. Material balances, an energy balance and Raoult's law for vapor-liquid equilibrium are used to determine the amounts of liquid and vapor exiting the drum and the mole fractions in each phase.
Details
Details
An overall and component mole balance are performed:
F
n
L
n
V
n
F
n
i
L
n
i
V
n
i
where is the number of moles, the superscripts , and refer to the feed, liquid and vapor streams and the subscript refers to a component (methanol or water).
n
F
L
V
i=morw
Overall and component energy balances with the reference state =25°C and =1bar:
T
ref
P
ref
H
in
H
out
F
H
L
H
V
H
F
H
i
L
H
i
V
H
i
where is enthalpy (kJ).
H
The enthalpies of each stream are calculated using heat capacities (kJ/(mol K)) and heat of vaporization (kJ/mol):
Cp
Δ
vap
H
F
H
F
n
i
L
Cp
i
T
in
T
ref
L
H
L
n
i
L
Cp
i
T
out
T
ref
V
H
V
n
i
V
Cp
i
T
out
T
ref
vap
H
i
The flash drum has a single equilibrium stage, so the exiting liquid and vapor streams are at the same temperature, .
T
out
Saturation pressure of the components in vapor-liquid equilibrium is calculated using the Antoine equation:
sat
P
sat
P
i
A
i
B
i
T+
C
i
where , and are Antoine constants for each component, and is in bar.
A
i
B
i
C
i
sat
P
Raoult's law is used for the exit streams to find the vapor-liquid equilibrium compositions:
x
i
sat
P
i
y
i
where and are the liquid and vapor mole fractions.
x
i
y
i
The sum of the mole fractions times their saturation pressures is the total pressure .
P=+
x
m
sat
P
m
x
w
sat
P
w
The screencast video at[1] explains how to use this Demonstration.
References
References
[1] Adiabatic Flash Drum with Binary Liquid Feed[Video]. (Dec 8, 2016) http://www.learncheme.com/simulations/separations/adiabatic-flash-drum-with-binary-liquid-feed.
Permanent Citation
Permanent Citation
Derek Machalek, Rachael L. Baumann, John L. Falconer
"Adiabatic Flash Drum with Binary Liquid Feed"
http://demonstrations.wolfram.com/AdiabaticFlashDrumWithBinaryLiquidFeed/
Wolfram Demonstrations Project
Published: June 18, 2015