Nernst Equation for Cellular Membranes
Nernst Equation for Cellular Membranes
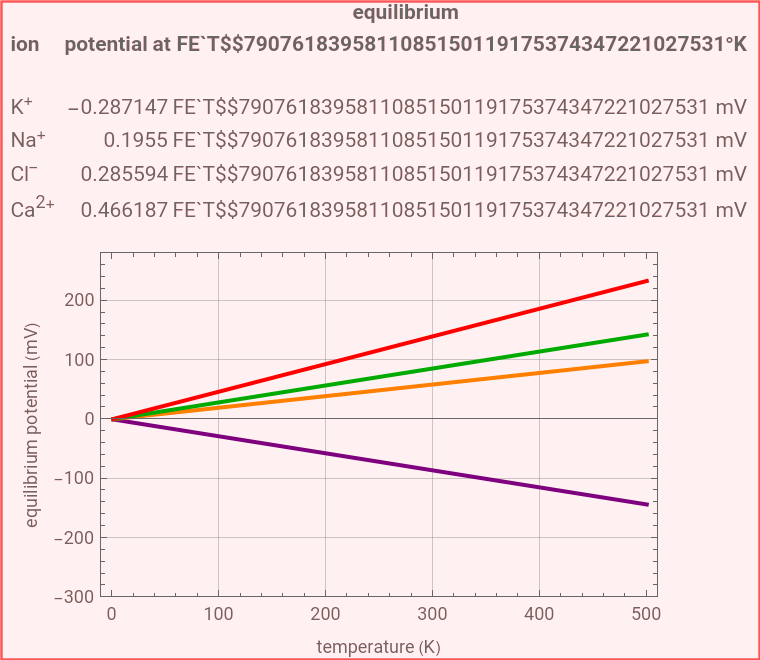
The Nernst equation is used to determine the potential of an ion of charge across a membrane using both extracellular and intracellular concentrations. It can be written as
z
E
m
RT
zF
[C]
i
[C]
o
RT
F
E
m
R=8.3144621
Joules
Kelvin·Mole
T
F=0.00096485339
Coulombs
Mole
[]
+
K
outside
[]
+
Na
outside
[]
-
Cl
outside
[]
2+
Ca
outside
and , , , and are the intracellular concentrations of potassium, sodium, chlorine, and calcium ions.
[]
+
K
inside
[]
+
Na
inside
[]
-
Cl
inside
[]
2+
Ca
inside
The default setting of the Demonstration calculates the equilibrium potentials for ions in a mammalian cell.
References
References
[1] D. H. Terman and G. B. Ermentrout, Foundations of Mathematical Neuroscience, New York: Springer, 2010 pp. 2–5.
External Links
External Links
Permanent Citation
Permanent Citation
Apoorva Mylavarapu
"Nernst Equation for Cellular Membranes"
http://demonstrations.wolfram.com/NernstEquationForCellularMembranes/
Wolfram Demonstrations Project
Published: January 1, 1999