Compressibility Factors Using the Soave-Redlich-Kwong Equation of State
Compressibility Factors Using the Soave-Redlich-Kwong Equation of State
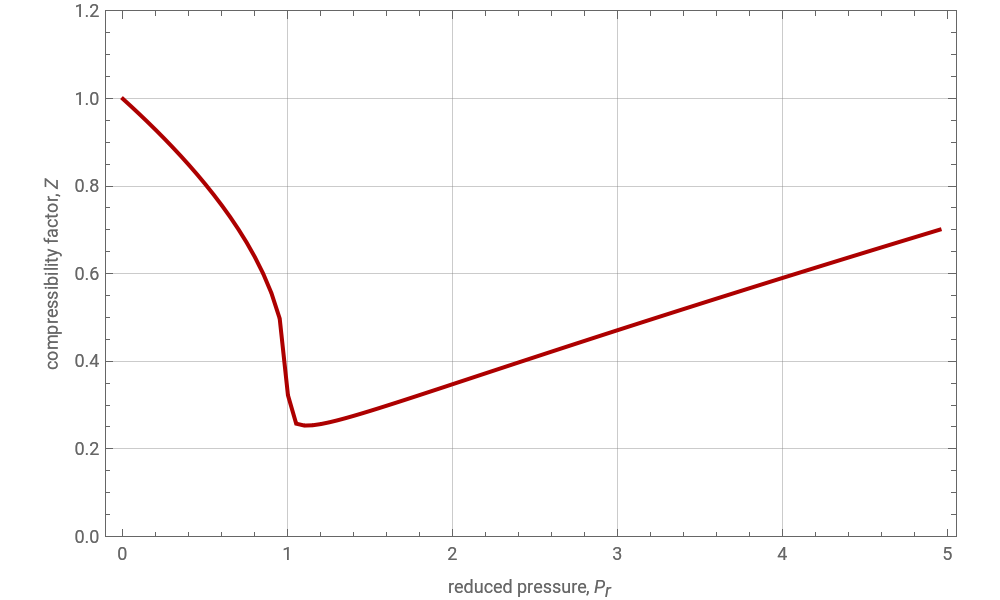
The Soave–Redlich–Kwong equation of state can accurately describe a compound in both liquid and vapor phases. This Demonstration applies this equation of state to estimate and plot the compressibility factor, , versus reduced pressure, =, for ethane at various values of the reduced temperature, =.
Z=
PV
RT
P
r
P
P
c
T
r
T
T
c
Details
Details
In the Soave–Redlich–Kwong equation of state, the compressibility factor is a solution of the following cubic equation:
f(Z)=-+(A-B-)Z-AB=0
3
Z
2
Z
2
B
where and with , , , and . Here is the acentric factor, and are the critical temperature and pressure, and = is the reduced temperature.
A=
aP
2
(RT)
B=
bP
RT
a=0.42748α
2
(R)
T
c
P
c
b=0.08664R/
T
c
P
c
α=
2
(1+m(1-))
T
r
m=0.480+1.574ω-0.176
2
ω
ω
T
c
P
c
T
r
T
T
c
For ethane, =305.43K, =48.84bar, and .
T
c
P
c
ω=0.09860
For more information on the compressibility factor, see http://en.citizendium.org/wiki/Compressibility_factor.
References
References
[1] Z. Nasri and H. Binous, "Applications of the Soave-Redlich-Kwong Equation of State Using Mathematica," Journal of Chemical Engineering of Japan, 40(6), 2007 pp. 534–538.
Permanent Citation
Permanent Citation
Housam Binous
"Compressibility Factors Using the Soave-Redlich-Kwong Equation of State"
http://demonstrations.wolfram.com/CompressibilityFactorsUsingTheSoaveRedlichKwongEquationOfSta/
Wolfram Demonstrations Project
Published: December 23, 2010