Thermokinetic Oscillator
Thermokinetic Oscillator
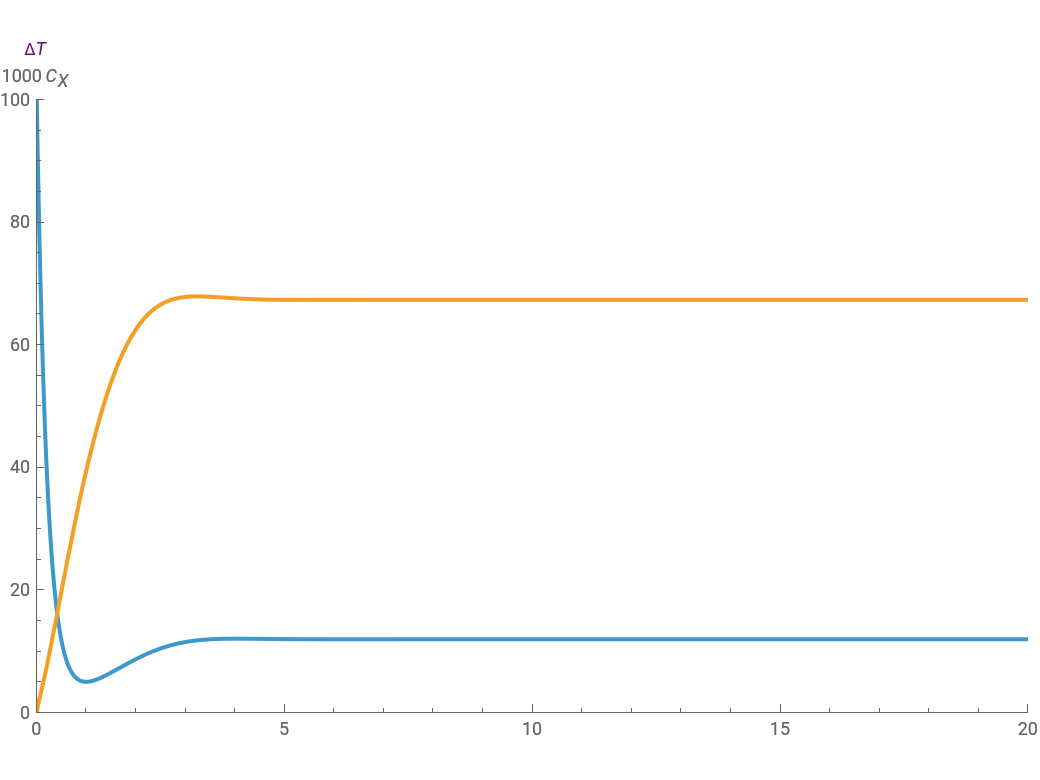
For a thermokinetic oscillator, the reaction scheme is , . The second reaction is exothermic with a reaction enthalpy ; it has a substantial activation energy . The concentration of component is constant, the temperature of the surroundings is , the density and specific heat of the reaction mixture are and , the area for heat transfer is , the volume of the system is , , , and . The relevant rate equations are solved and plotted for and , the latter scaled up by 1000 for convenience.
A⟶X
X⟶R
H
E
A
T
0
ρ
c
ρ
A
V
α=
2
(E/(RT))
β=Ah(Vρ)
c
ρ
γ=H(ρ)
k
2
T
0
c
ρ
ΔT(t)
C
X
Details
Details
The reaction produces heat, accelerating the reaction and reducing the concentration of . As you vary the parameters, the interplay of these opposing tendencies can produce oscillations and bifurcation behavior.
C
X
References
References
[1] P. Gray and S. K. Scott, Chapter 4, Chemical Oscillations and Instabilities, New York: Oxford University Press, 1990.
[2] D. A. Frank-Kamenetskii and I. E. Salnikov. Zhur. Fiz. Khim., 17, 1943 p. 79.
[3] xmds.org. thermkin.xmds. (Dec 28 2012)
Permanent Citation
Permanent Citation
Benson R. Sundheim
"Thermokinetic Oscillator"
http://demonstrations.wolfram.com/ThermokineticOscillator/
Wolfram Demonstrations Project
Published: January 11, 2012